Global Frontotemporal Disorders Treatment Market Size Value Analysis 2022-2029 by Shubham Channe - Issuu

Selective Chemical Oxidation of Risperidone: A Straightforward and Cost‐Effective Synthesis of Paliperidone - Riva - 2011 - European Journal of Organic Chemistry - Wiley Online Library
Package leaflet: Information for the user Paliperidone Teva 25 mg Prolonged-release Suspension for Injection Paliperidone Teva 5
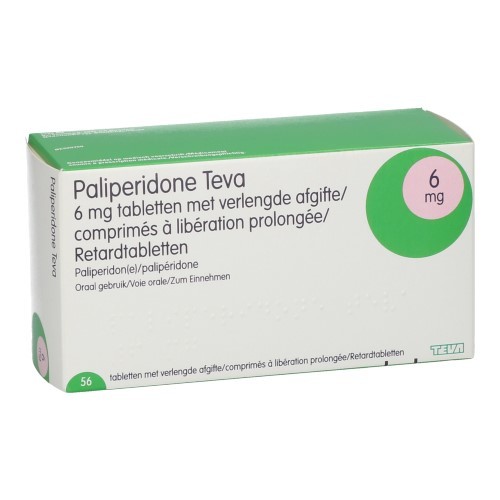
Gebrauchsinformation: Information für Patienten Paliperidone Teva 3 mg Retardtabletten Paliperidone Teva 6 mg Retardtabletten P
Teva and MedinCell Looking to Enter Increasingly Competitive LAI Schizophrenia Market - Clinical Trials Arena

PDF) Switching from oral risperidone to flexibly dosed oral paliperidone extended-release: Core symptoms, satisfaction, and quality of life in patients with stable but symptomatic schizophrenia: The RISPALI study
Package leaflet: Information for the user Paliperidone Teva 25 mg Prolonged-release Suspension for Injection Paliperidone Teva 5

Risperidone teva generics 1 mg compresse rivestite con film - 4 mg compresse rivestite con film 60 compresse
Dissolution Test Failure Leads to Teva Schizophrenia Product Recall - Pharmaceutical Processing World

PDF) Once-monthly paliperidone palmitate compared with conventional and atypical daily oral antipsychotic treatment in patients with schizophrenia

US9439906B2 - Dosing regimen associated with long acting injectable paliperidone esters - Google Patents