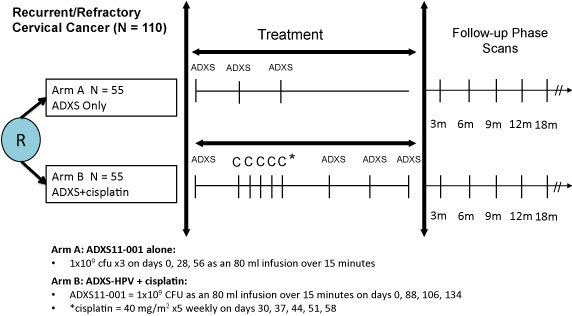
PDF) A Randomized Phase 2 Study of ADXS11-001 Listeria monocytogenes–Listeriolysin O Immunotherapy With or Without Cisplatin in Treatment of Advanced Cervical Cancer

PDF) Systemic listeriosis following vaccination with the attenuated Listeria monocytogenes therapeutic vaccine, ADXS11-001

PDF) A Randomized Phase 2 Study of ADXS11-001 Listeria monocytogenes–Listeriolysin O Immunotherapy With or Without Cisplatin in Treatment of Advanced Cervical Cancer
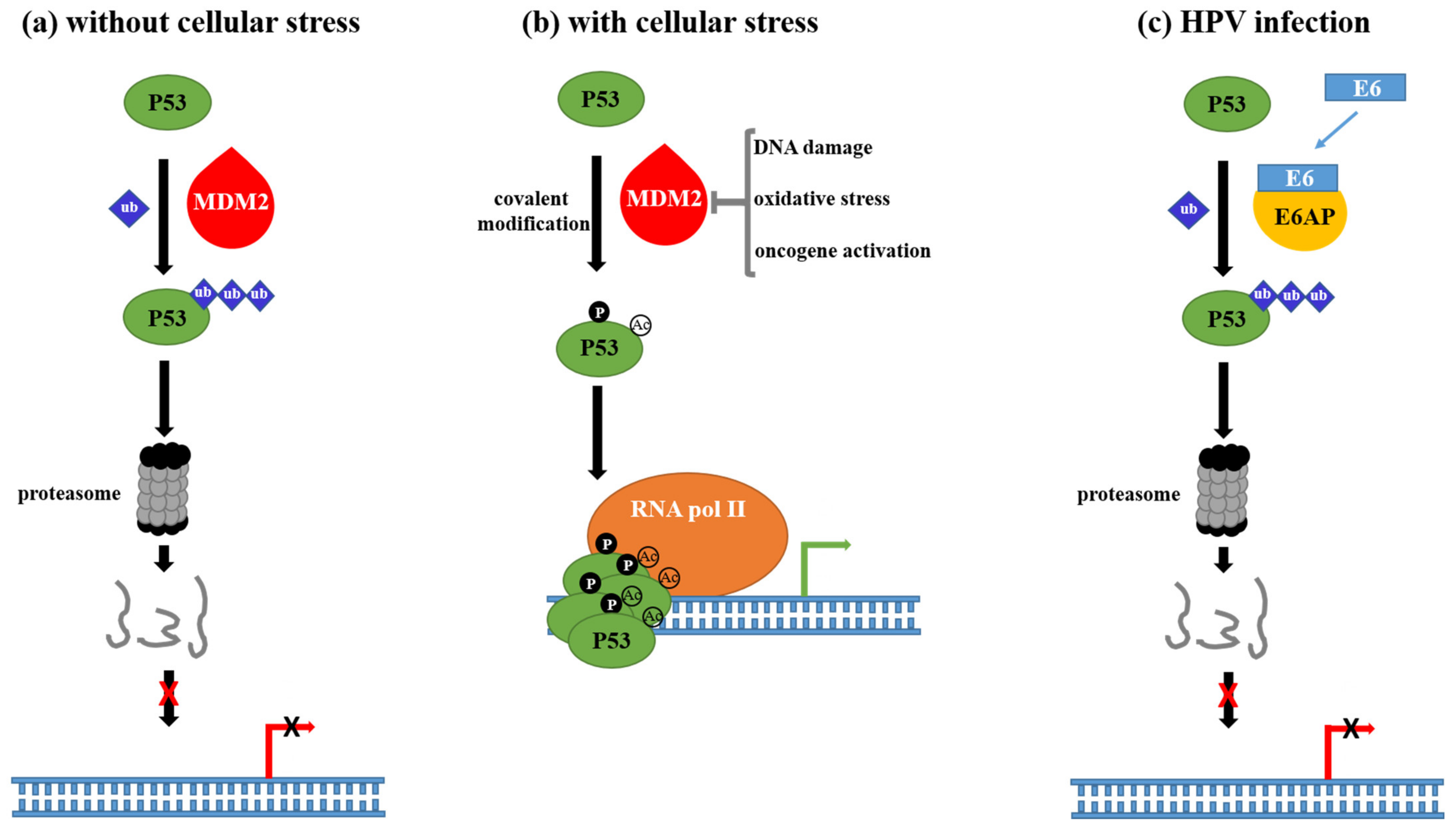
Cells | Free Full-Text | The Interaction of Human Papillomavirus Infection and Prostaglandin E2 Signaling in Carcinogenesis: A Focus on Cervical Cancer Therapeutics | HTML
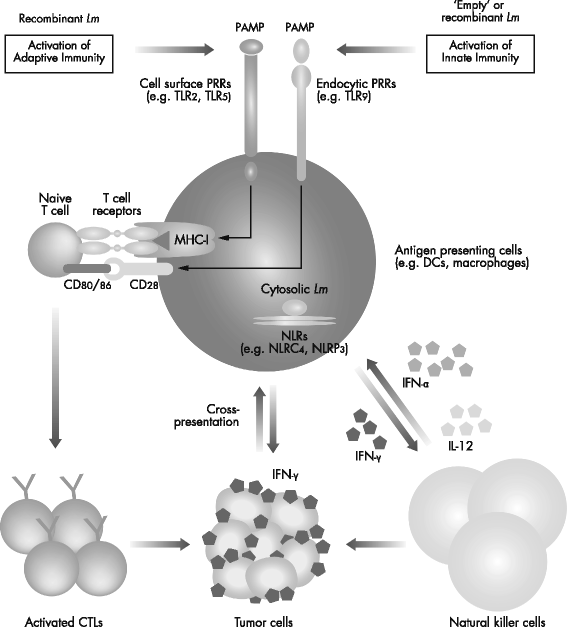
Mechanistic insights into ADXS11-001 human papillomavirus-associated cancer immunotherapy | Gynecologic Oncology Research and Practice | Full Text

Personalized cancer vaccination in head and neck cancer - Shibata - 2021 - Cancer Science - Wiley Online Library
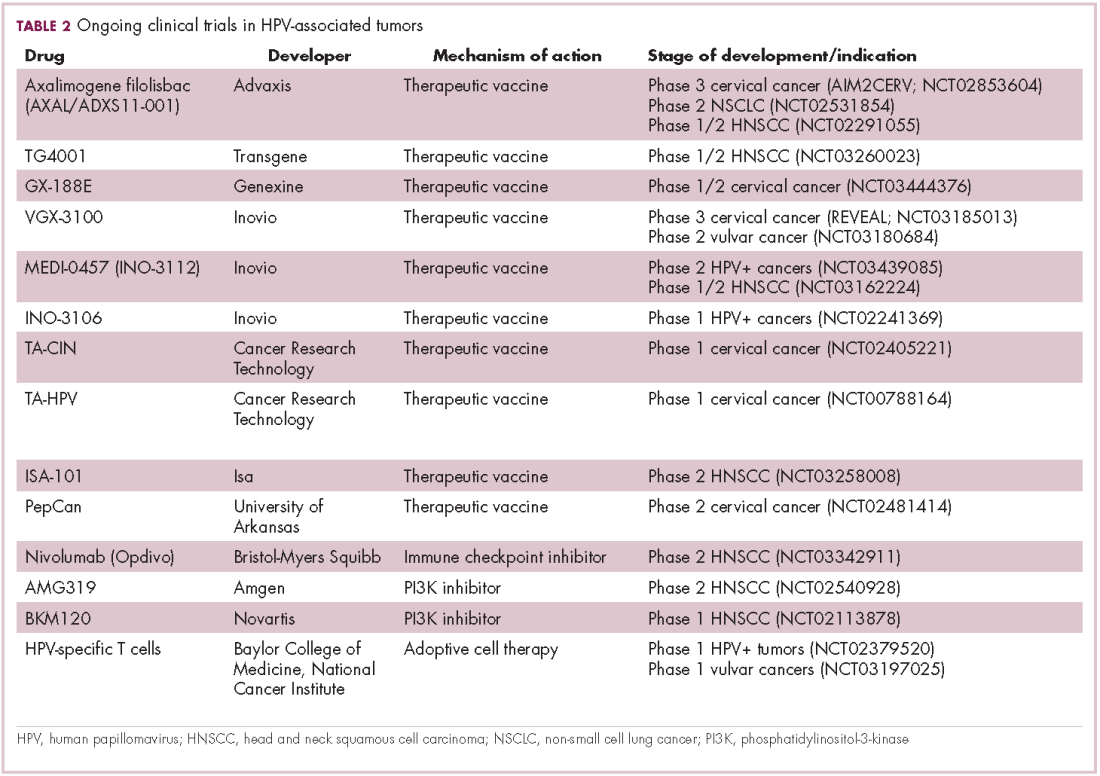
Immunotherapy may hold the key to defeating virally associated cancers | MDedge Hematology and Oncology
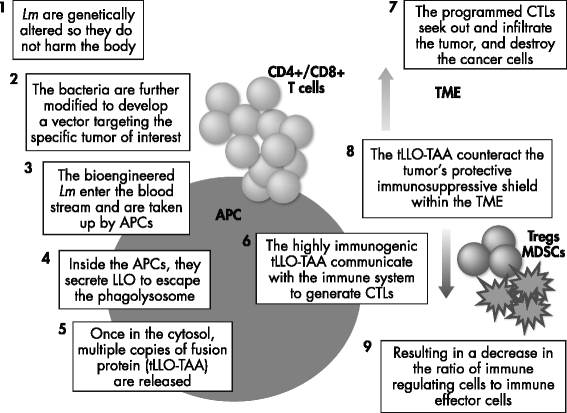
Mechanistic insights into ADXS11-001 human papillomavirus-associated cancer immunotherapy | Gynecologic Oncology Research and Practice | Full Text
ADXS11-001 immunotherapy in squamous or non-squamous persistent/recurrent metastatic cervical cancer: Results from stage 1 [and

Frontiers | Clinical Experience and Recent Advances in the Development of Listeria-Based Tumor Immunotherapies

Phase II study of axalimogene filolisbac (ADXS-HPV) for platinum-refractory cervical carcinoma: An NRG oncology/gynecologic oncology group study - ScienceDirect

Beyond Chemotherapy: An Overview and Review of Targeted Therapy in Cervical Cancer. | Semantic Scholar

The Role of Biomarkers for the Prediction of Response to Checkpoint Immunotherapy and the Rationale for the Use of Checkpoint Immunotherapy in Cervical Cancer - Clinical Oncology
ADXS11-001 immunotherapy in squamous or non-squamous persistent/recurrent metastatic cervical cancer: Results from stage 1 [and



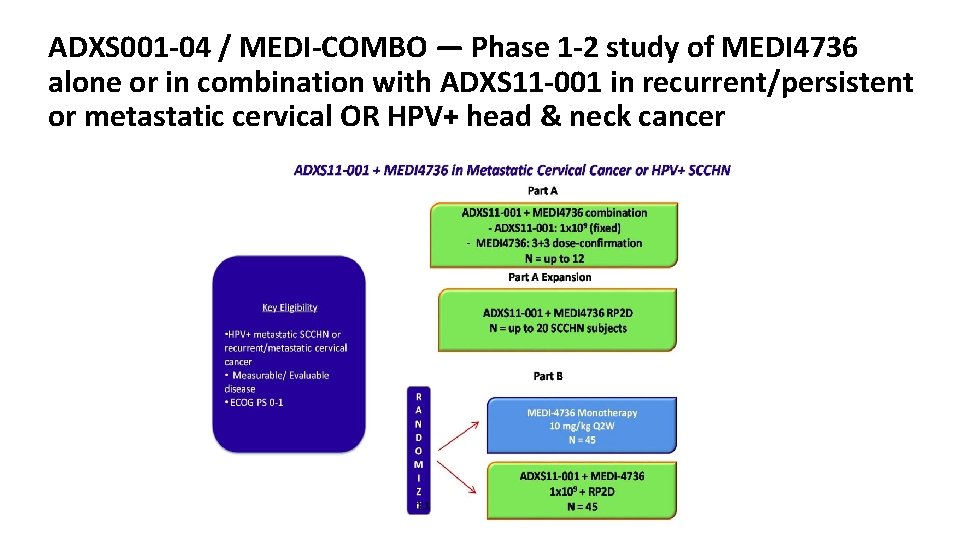
:%0A%0ANivolumab%20for%20Oral%20Squamous%20Cell%20Carcinoma.png?md=1)
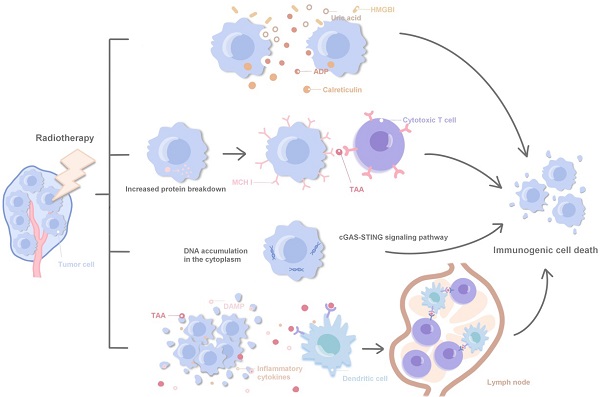



%20for%20Carcinoma,%20Squamous%20Cell.png?md=1)